The Contribution of Calcium to Changes in Leaf Anatomical Character of Oil Palm Seedlings (Elaeis guineensis Jacq.) under Drought Stress
Novi Yulanda Sari(1*), Eka Tarwaca Susila Putra(2)
(1) Faculty of Agriculture Universitas Gadjah Mada, Yogyakarta
(2) Faculty of Agriculture Universitas Gadjah Mada, Yogyakarta
(*) Corresponding Author
Abstract
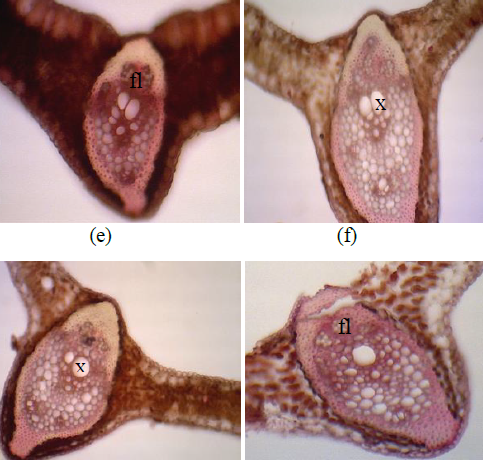
The research was purposed (1) to know the effects of drought stress on changes in leaf anatomical character of oil palm seedlings (2) to know the contribution of calcium in cell compactness and increase the structural strength of leaf tissue so that oil palm seedlings were more tolerant to drought stress. This experiment was laid out following a split plot design with three blocks as replication. Main plot consists of drought stress levels, that are field capacity (FTSW 1.00), moderate drought stress (FTSW 0.35) and severe drought stress (FTSW 0.15). The fraction of transpirable soil water (FTSW) is a method for evaluating gradually increasing drought stress based on the amount of water loss due to transpiration. Meanwhile, subplot consists of four doses of calcium (Ca), that are of 0.0 g/seedlings, 0.04 g/seedlings, 0.08 g/seedlings, and 0.12 g/seedlings. Calcium fertilizer used is calcium sulfate (CaSO4) pure analysis. Leaf anatomical character was observed including the epidermal length and epidermal width; hypodermal length and hypodermal width; palisade cell length and palisade cell width; sponge cell length and sponge cell width; mesophyll tissue thickness; xylem and phloem diameter. The results showed that moderate and severe drought stress reduced epidermal cell length, upper hypodermal cell width, mesophyll thickness, palisade width and phloem diameter of leaf vessels. The applications of calcium to the leaf of oil palm seedlings under drought stresses were able to increased in the sponge cell length at a Ca dosage of 0.04 g/seedlings; increased lower hypodermal width and diameter phloem at a Ca dosage of 0.04 g/seedlings; and increased diameter xylem of leaves vessel at a Ca dosage of 0.12 g/seedlings.
Keywords
Full Text:
PDFReferences
Boughalleb, F., R. Abdellaoui, N. Ben-Brahim and M. Neffati. 2014. Anatomical Adaptations of Astragalus gambiformis Pomel. under Drought Stress. Central European Journal of Biology, 9(12), pp. 1215-1225.
Burnett, S. E., S. V. Pennisi, P. A. Thomas and M. W. van Iersel. 2005. Controlled Drought Affects Morphology and Anatomy of Salvia splendens. J. Amer. Soc. Hort. Sci., 130, pp.775-781.
Chartzoulakis, K., A. Patakas, G. Kofindis, A. Bosabalidis and A. Nastou. 2002. Water Stress Affect Leaf Anatomy, Gas Exchange, Water Relations and Growth of Two Avocado Cultivars. Scientia Horticulture, 95, pp. 39-50.
Easterwood. 2002. Calcium's Role in Plant Nutrition. Fluid Jurnal.
Ennajeh, M., A. M. Vadel, H. Cochard and H. Khemira. 2010. Comparative Impacts of Water Stress on the Leaf Anatomy of a Drought-Resistant and a Drought Sensitive Olive Cultivar. Journal of Horticultural Science & Biotechnology, 85(4) pp. 289-294.
Esau, K. 1977. Anatomy of Seed Plant2nd Ed. Printed in the United States of America, 125-130.
Fairhurst, T. and D. McLaughlin. 2009. Sustainable Oil Palm Development on Degraded Land in Kalimantan. World Wildlife Fund.
Gardner, F. P., R. B. Pearce and G. L. Mitchell. 1991. The Physiology of Cultivated Plants (Fisiologi Tanaman Budidaya, alih bahasa H. Susilo). Jakarta: Universitas Indonesia Press, 421 p.
Hepler, P. K. 2005. Calcium: A Central Regulator of Plant Gowth and Development. Plant Cell, 17(8), pp. 2142-2155.
Kutlu, N., R. Terzi, C. Tekeli, G. Senel, P. Battal and A. Kadioglu. 2009. Changes in Anatomical Structure and Dosages of Endogenous Phytohormones during Leaf reallIng in Ctenanthe setosa under Drought Stress. Turk J. Biol, 331(8).
Laurance, W. F. 2007. Forest deStruction in Tropical Asia. Current science, 93(11), pp. 1544-1550.
Lovisolo, C. and A. Schubert. 1998. Effects of Water Stress on Vessel Size and Xylem Hydraulic Conductivity in Vitis vinifera L. Journal of Experimental Botany, 49, pp. 693-700.
Mardiyah, S. 2014. Pengaruh Kekeringan dan Pupuk Organik terhadap Anatomis dan Pertumbuhan Tanaman Padi Gogo (Oriza sativa L. ‘ Situ Bagendit') pada Tanah Berkapur. Tesis. Universitas Gadjah Mada: Yogyakarta.
Masinde, P. W., H. Stutzel and A. Fricke. 2006. Plant growth, water relations and
transpiration of two species of African nightshade (Solanum villosum Mill. ssp.
Miniatum (Bernh. ex Willd.) edmonds and S. sarrachoides Sendtn.) under
water-limited conditions. Scientia Horticulturae 110: 7–15.
Muis, A. 2016. Perubahan Aktivitas Biokimiawi dan Fisiologis Empat Progeni Kelapa Sawit (Elaeis guineensis Jacq.) pada Berbagai Aras Lengas Tanah. Tesis. Universitas Gadjah Mada: Yogyakarta.
Naeem, M., M. N. Khan, M. N. A. Khan and Moinuddin. 2013. Adverse Effects of Abiotic Stresses on Medicinal and Aromatic Plants and Their Alleviation by Calcium. Springer Science+Business Media New York.
Patakas, A., N. Nikolaou, E. Zioziou, K. Radoglou and B. Noitsakis. 2002. The Role of Organic Solute and Ion Accumulation in Osmotic Adjustment in Drought Stressed Grapevines. Plant Sci, 163, pp. 361-367.
Pena-Valdivia, C. B., A. B. Sanchez-Urdaneta, J. M. Ranegl, J. J. Munoz, R. Garcia-Nava and R. C. Velazquez. 2010. Anatomical Root Variations in Response to Water Deficit: Wild and Domesticated Common Bean (Phaseolus vulgaris L.). Biol. Res, 43, pp. 417-427.
Pratiwi, R. 2014. Mengenal sifat tanah masam gambut dan tanah masam Ultisol.
Balai Besar Pelatihan Pertanian Lembang : Jawa Barat.
Ray J. D and T. R. Sinclair. 1998. The Effect of Pot Size on Growth and Transpiration of Maize and Soybean during Water Deficit Stress. J Exp Bot, 49, pp. 1381-1386.
Robertson, D. 2013. Modulating Plant Calcium for Better Nutrition and Stress Tolerance. Botany, Vol 2013, 22 p.
Rosawanti, P., M. Ghulamandhi and N. Khumaida. 2015. Respon Anatomi dan Fisiologi Akar Kedelai Tercekam Kekeringan. Jurnal Agronomi Indonesia, 43(3), pp. 186-192.
Shao, H., L. Chu, C. A. Jaleel and C. Zhao. 2008. Water deficit stress induced anatomical changes in higher plants. C. R. Biologies, 331 (2008), pages 215-225.
SMARTRI. 2014. SOP of experimental nursery.
Song, W. Y., Z. B. Zhang, H. B. Shao, X. L. Guo, H. X. Cao, H. B. Zhao, Z. Y. Fu and H. J. Hu. 2008. Relationship between Calcium Decoding Elements and Plant Abiotic Stress Resistance. Int J Biol Sci, 4, pp. 116-125.
Stibig, H. J., F. Achard, S. Carboni, R. Rasi and J. Miettinen. 2014. Change in Tropical Forest Cover of Southeast Asia from 1990 to 2010. Biogeosciences, 11, pp. 247-258.
Subagyo, H., N. Suharta, dan Agus. B. Siswanto. 2000. Sumber daya Lahan Indonesia dan Pengelolaannya. Pusat Penelitian Tanah dan Agroklimat : Bogor, pp. 21-66.
Taiz, L. and E. Zeiger. 2002. Plant Physiology. Third edition. Sinauer Associates Inc. P. 67, 70.um. Annals of Botany, 883, pp. 55-360.
Tardieu, F. 2000. Drought Perseption by Plants do Cells of Droughted Plants Experiences Water Stress? The Diversity of Adaptation in the Wide. Plant Gowth Regulation, 20, pp. 93-104.
Tim Peneliti Ilmu Tanah dan Agronomi. 2016. Fakta tentang Patah Pelepah pada Kelapa Sawit. PPKS note edisi Maret. Medan: Pusat Penelitian Kelapa Sawit.
Wu, Y. and D. J. Cosgove. 2000. Adaptation of Root Low Water Potensials by Changes in Cell Wall Extensibility and Cell Wall Proteins. J. Exp. Bot, 51, pp. 1543-1553.Article Metrics
Refbacks
- There are currently no refbacks.
Ilmu Pertanian (Agricultural Science) ISSN 0126-4214 (print), ISSN 2527-7162 (online) is published by Faculty of Agriculture Universitas Gadjah Mada collaboration with Perhimpunan Sarjana Pertanian Indonesia (PISPI) and licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.











_2025_-_kecil_.png)
_2024_kecil_2.png)
