ADSORPSI POLUTAN ION DIKROMAT MENGGUNAKAN ZEOLIT ALAM TERMODIFIKASI AMINA (Adsorption of Dichromate Ions Pollutant Using Ammine Modified-Natural Zeolites)
Eko Sri Kunarti(1*), Sutarno Sutarno(2), Sampe Baralangi(3)
(1) Departemen Kimia, Fakultas Matematika dan Ilmu Pengetahuan Alam, Universitas Gadjah Mada, Sekip Utara, Yogyakarta, 55281.
(2) Departemen Kimia, Fakultas Matematika dan Ilmu Pengetahuan Alam, Universitas Gadjah Mada, Sekip Utara, Yogyakarta, 55281.
(3) Madrasah Aliyah Negeri Makale, Jl. Tritura 188 Makale, Tana Toraja, 91819.
(*) Corresponding Author
Abstract
ABSTRAK
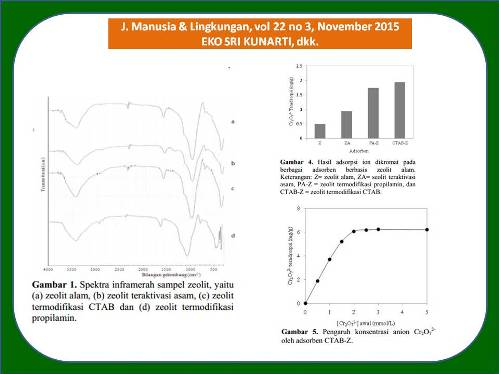
Kromium (VI) merupakan polutan logam berat berbahaya bagi kesehatan dan lingkungan oleh karena itu pengambilan ion Cr(VI) dalam air penting dilakukan untuk mengatasi pencemaran lingkungan. Proses adsorpsi merupakan salah satu teknik sederhana yang dapat digunakan untuk pengambilan ion logam. Pada penelitian ini telah dilakukan kajian adsorpsi ion dikromat sebagai model limbah Cr(VI) dalam air menggunakan adsorben zeolit alam termodifikasi amina. Penelitian diawali dengan preparasi adsorben zeolit alam termodifikasi amina. Preparasi dimulai dengan pencucian zeolit alam menggunakan akuades, kemudian refluks zeolit alam menggunakan HCl 3M. Zeolit hasil refluks selanjutnya dimodifikasi menggunakan garam ammonium kuarterner, N-cethyl-N,N,N-trimethylammonium bromide (CTAB) dan amina primer, propilamin (PA). Zeolit alam (Z), zeolit teraktivasi asam (ZA) dan zeolit hasil modifikasi amina selanjutnya digunakan sebagai adsorben untuk adsorpsi anion dikromat. Karakterisasi adsorben dilakukan dengan mengunakan metode spektroskopi infaramerah dan difraksi sinar-X, sedangkan jumlah anion dikromat yang teradsorpsi dianalisis dengan spektroskopi serapan atom. Hasil penelitian menunjukkan bahwa sampel zeolit mengandung mineral klinoptilolit, mordernit dan kuarsa. Struktur zeolit tidak mengalami kerusakan oleh perlakuan termal dan perlakuan kimia. Modifikasi zeolit meningkatkan efisiensi adsorpsi zeolit alam. Ion dikromat dapat teradsorpsi dengan lebih baik oleh zeolit termodifikasi amina daripada zeolit teraktivasi asam dan zeolit tanpa modifikasi, dengan kemampuan adsorpsi zeolit termodifikasi CTAB (CTAB-Z) lebih besar daripada zeolit termodifikasi propilamin (PA-Z). Adsorpsi ion dikromat pada adsorben zeolit berlangsung baik dengan urutan CTAB-Z > PA-Z > ZA > Z, dengan kemampuan adsorpsi masing-masing sebesar 1,96; 1,74; 0,90 dan 0,48 mg/g. Adsorpsi anion dikromat oleh zeolit termodifikasi CTAB merupakan adsorpsi kimia (kemisorpsi) dengan energi adsorpsi sebesar 24,66 kJ/mol.
ABSTRACT
Chromium (VI) is a heavy metal pollutant that is harmful to health and the environment, therefore Cr(VI) ions in aqueous solution removal is important to overcome the environmental pollution. Adsorption process is one of simple techniques that can be used to take metal ions. Adsorption study of dichromate ions as a wastewater model of Cr(VI) has been conducted in this research by using ammine modified natural zeolite as adsorbents. The research was initiated by preparation of adsorbent of ammine-modified natural zeolites. The preparation was started by washing the natural zeolite (Z) using distilled water and refluxing by 3M hydrochloric acid. Refluxed zeolites (ZA) were modified by a quaternary ammonium salt, N-cethyl-N,N,N-trimethylammonium bromide (CTAB), and a primary amine, propylamine (PA). The natural zeolite (Z), acid activated zeolite (ZA) and amine-modified zeolites were then applied for adsorption of dichromate ions. Characterization of zeolite samples was performed by infrared spectroscopy and X-ray diffraction methods to confirm the crystal structure, and atomic absorption spectroscopy method to analyze the adsorbed dichromate ions. Results showed that all zeolite samples contain clinoptilolite, mordernite and quartz minerals. The zeolite structure was not changed by heat and chemical treatments. Modification of zeolites enhanced the adsorption efficiency of natural zeolites. The dichromate ions were better adsorbed on ammine modified-zeolites compared to that of unmodified-zeolite with adsorption ability of CTAB modified zeolite (CTAB-Z) was greater than that of propylammine modified zeolite (PA-Z). The adsorption of dichromate on zeolite samples was achieved in the order of CTAB-Z > PA-Z > ZA > Z, with the adsorption efficiency was about 1.96; 1.74; 0.90 and 0.48 mg/g, respectively. The dichromate ions adsorption by CTAB modified zeolite is chemical adsorption (chemisorption) with the adsorption energy of 24.66 kJ/mol.
Keywords
Full Text:
Artikel lengkap (PDF) (Bahasa Indonesia) Gambar 1 (Bahasa Indonesia) Gambar 2 (Bahasa Indonesia) Gambar 3 (Bahasa Indonesia)References
Adamson, A.W., 2004. Physical Chemistry of Surfaces. 5th edition. John Wiley and Sons. New York.
Bajpai, A.K., dan Rai, L., 2010. Removal of Chromium Ions from Aqueous Solution by Biosorption on to Ternary Biopolymeric Microspheres. Indian Journal of Chemical Technology, 17:17-27.
Chidambaram, A., Sundaramoorthy, P., Murugan, A., Ganesh, K.S., dan Baskaran, L., 2009. Chromium Induced Cytotoxicity in Blackgram (Vigna Mungo L.). Iran. J. Environ. Health Sci. Eng., 6(1):17-22.
Chmielewska, E., 2003. Adsorption of Arsenate and Chromate from Waters on Hydrophobized Zeolitic Media. Turk. J. Chem., 27:639 -648.
Erdem, M., Altundogan, H.S., dan Tumen, F., 2004. Removal of Hexavalent Chromium by Using Heat-Activated Bauxite. Mineral Engineering, 17:1045-1052.
Ghadiri, S. K., Nabizadeh, R., Mahvi, A. H., Nasseri, S., Kazemian, H., Mesdaghinia, A. R., dan Nazmara, S.H., 2010. Methyl Tert-Butyl Ether Adsorption on Surfactant Modified Natural Zeolites. Iran. J. Environ. Health Sci. Eng., 7(3):241-252.
Ghiaci, M., Abbaspur, A., Kia, R., dan Seyedeyn-Azad, F., 2004. Equilibrium Isotherm Studies for the Sorption of Benzene, Toluene, and Phenol onto Organo Zeolite and As-Synthesized MCM-41. Separation and Purification Technology 40:217–229.
Gopalakrishnan, S., Kannadasan, T., Velmurugan, S., Muthu, S., dan Kumar, V.P., 2013. Biosorption of Chromium (VI) from Industrial Effluent using Neem Leaf Adsorbent. Research Journal of Chemical Sciences, 3(4):48-53.
Gupta, V.K., dan Rastogi, A., 2009. Biosorption of Hexavalent Chromium by Raw and Acid-Treated Green Alga Oedogonium Hatei from Aqueous Solutions. J. Hazard Mater., 163: 396-402.
Hemalatha P.V., dan Prasada R.P.V., 2012. Adsorption of Cr (VI) Ions from Aqueous Solutions Using Calcined Brick. International Journal of Environmental Sciences, 2(4):2321-2327.
Kuleyin, A., 2007. Removal of Phenol and 4-Chlorophenol by Surfactant-Modifed Natural Zeolite. J. Hazard. Mater., 144:307–315.
Li, Z., dan Bowman, R.S., 2001. Retention of Inorganic Oxyanions by Organo-Kaolinite. Water Research, 35(16):3771–3776.
Li, Z., Burt, T., dan Bowman, R.S., 2000. Sorption of Ionizable Organic Solutes by Surfactant-Modified Zeolite. Environmental Science and Technology, 34:3756–3760.
Mortuza, M.G., Takahashi, T, Ueki, T., Kosaka, T., Michibata, H., dan Hosoya, H., 2005. Toxicity and Bioaccumulation of Hexavalent Chromium in Green Paramecium, Paramecium bursaria. J. Health Sci., 51(6), 676-682.
Popov, N., Popova, T., Rubio, J., dan Taffarel, S.R., 2012. Use of Natural and Modified Zeolites from Bulgarian and Chilian Deposits to Improve Adsorption of Heavy Metals from Aqueous Solutions. Bulletin of Mineralogy Petrology and Geochemistry, 49:83-93.
Roto, R., Tahir, I., dan Solikhah, U.N., 2009. Aplikasi Pengolahan Polutan Anion Khrom(VI) dengan Agen Penukar Ion Hydrotalcite Zn-Al-SO4. Jurnal Manusia dan Lingkungan, 16(1):41-53.
Taffarel, S.R., dan Rubio, J., 2010. Adsorption of Sodium Dodecyl Benzene Sulfonate from Aqueous Solution Using a Modified Natural Zeolite with CTAB. Minerals Engineering, 23:771–779
Vujakovic, A.D., Magdalena, R., Tomasevic-Canovic, M.R., Dakovic, A.S., dan V.T. Dondur, V.T., 2000. The Adsorption of Sulphate, Hydrogen Chromate and Dihydrogen Phosphate Anions on Surfactant-Modified Clinoptilolite. Applied Clay Science, 17:265–277.
Article Metrics
Refbacks
- There are currently no refbacks.
Copyright (c) 2017 Jurnal Manusia dan Lingkungan







